

In remote areas, treatable diseases like sickle cell anemia and malaria have a devastating impact mainly because access to early detection and treatment is limited. Most people need help to travel from their remote villages to get tested in big cities where test labs and diagnostics are available. For those living outside the reach of these labs, there is typically no testing available and, therefore, no early treatment. At the time, costly and time-consuming laboratory testing was the only path for confirming a diagnosis.

These challenges drove Portland innovator Hemex Health to develop in-the-field testing solutions in 2017. Hemex had already secured startup funding to develop technologies with Cleveland’s Case Western Reserve University to commercialize a new blood analysis technology: the HemeChip. The HemeChip would need to deliver multiple solutions to make remote diagnostic blood testing viable: it had to be low-cost, compact, portable, and provide rapid analysis.
With millions of dollars of funding at stake, Hemex needed an engineering partner with experience in accelerated R&D and MedTech device prototyping. Hemex engaged with AC to engineer the rapid prototyping of a medical device that could enter the field as soon as possible. They needed a compact, ruggedized solution within a short development window and limited budget. Our Product Development engineers were eager to help them design the vehicle for their emerging HemeChip technology and realize their vision of what was called a “test lab in a box.”

There are definitely countries in Africa where 25% of all child mortality is being caused by sickle cell disease. And if you can diagnose them, 70% of those kids would live. And if they’re not diagnosed, more than half die by their 5th birthday. So we’re talking 750 kids a day are dying from sickle cell disease. -Patti White, CEO & Co-Founder of Hemex Health
As we partnered closely with Hemex, we became an integrated extension of their team, deploying our mechanical, electrical, and software engineers to tackle the project’s urgent goals:
The first and most critical milestone was to deliver a field-testable prototype capable of validating Hemex’s detection method in real-world settings. Designed for deployment in Africa and India, the HemeChip was used to gather efficacy data and prepare for a multi-phase clinical trial.
Guided by aggressive development timelines and budget constraints, we prioritized design efficiency. Wherever possible, we leveraged off-the-shelf components and minimized customization without compromising the functionality required for field deployment. These strategic decisions enabled us to quickly deliver a rugged, working device into the hands of global research teams.

HemeChip Portable Test Lab Prototype
Designing for high-voltage medical applications is just one of our core competencies, and it was central to the success of the HemeChip prototype. The device needed to deliver either a fixed voltage or current within a highly specific range (100–500V and 0–12 mA) based on values determined in the lab prior to field deployment. Our engineers developed a user-controlled interface that allowed operators to toggle between voltage and current modes, with an on-screen slider enabling real-time adjustments and readouts. This enabled principal investigators to fine-tune electrical settings in advance while ensuring reliable consistency during clinical use in the field.
Ease of use was critical for a diagnostic device that would be handled by both trained lab personnel and a global team of field researchers who would have only minimal training. Our team prioritized simplicity, designing an intuitive user experience that reduced cognitive load and streamlined operation. The result was an interface that could be learned quickly and operated reliably across a wide range of technical backgrounds, without sacrificing functionality or control.
Because HemeChip diagnoses are performed through offline image processing, image clarity and consistency were mission-critical. Leveraging our deep background in imaging system design, we ensured the prototype captured high-resolution, low-distortion images under variable field conditions. This fidelity was key to enabling accurate downstream interpretation and ultimately supporting confidence in the device’s clinical value.
To support the needs of both research scientists and field technicians, we created a dual-mode interface that tailored the user experience to the context of use. The field mode featured a simple “one big button” design to minimize training requirements and reduce the chance of user error. Wheras, the lab mode unlocked full system functionality for configuration, calibration, and advanced control. This dual approach reflected our experience developing interfaces for life-critical systems, where both simplicity and precision must coexist.
Real-Time Electrical Control - An on-screen slider enabled precise voltage or current adjustments for testing blood samples. This intuitive interface helped researchers reliably set parameters and confirm output on the spot.
Built-In Safety Architecture - To mitigate high-voltage risks, we implemented a multilayered safety interlock system—ensuring operator protection without slowing down use in the field.
High-Fidelity Image Capture - Because diagnosis relied on offline image processing, the optics were tuned for consistent, high-resolution images—critical for accurate interpretation and reduced retests.
Ruggedized for Real-World Conditions - Built for field use, the prototype featured a hardened enclosure, reinforced internals, and stable architecture to withstand transport, handling, and environmental stress.
Although the initial requirements for the device were outlined over the phone and were very rudimentary, as development proceeded, AC made discoveries that refined the requirements and improved the definition of testing, as well as how the samples should be prepared. All of this information was then fed back to the researchers, allowing them to incorporate our findings into their protocol, resulting in a win-win for all parties.
We also collected valuable user feedback in the field with the HemeChip prototype that informed interative design decisions and ongoing refinements. The success of the first units deployed in the field also confirmed the need for rapid production of additional devices to fully equip the rest of the field team and expand Hemex's scope of testing.

Image Credit: Hemex Health | HemeChip Prototype Training at EHealth Africa in Nigeria (2018)
AC delivered more than just functional prototypes. We enabled Hemex Health to take a critical step toward real-world clinical validation. By assembling a multi-disciplinary team of mechanical, electrical, and software engineers, we rapidly transformed concept requirements into a rugged, field-ready diagnostic device, despite aggressive time and budget constraints.

Image Credit: Hemex Health | HemeChip Prototype Testing in Nigeria (2017)
“We were looking for innovation that solved big problems, had proven clinical results, could be developed in 2 years and had a strong business model that would attract investors.” - Hemex Health CEO and Co-Founder, Patti White
Our engineering partnership culminated in the successful production of 10 working units, empowering researchers to initiate global field testing and gather essential data for clinical progression. This rapid prototyping effort not only validated the core technology but also laid the foundation for future development phases—proving that the right engineering team can turn bold MedTech ideas into tangible, testable outcomes.
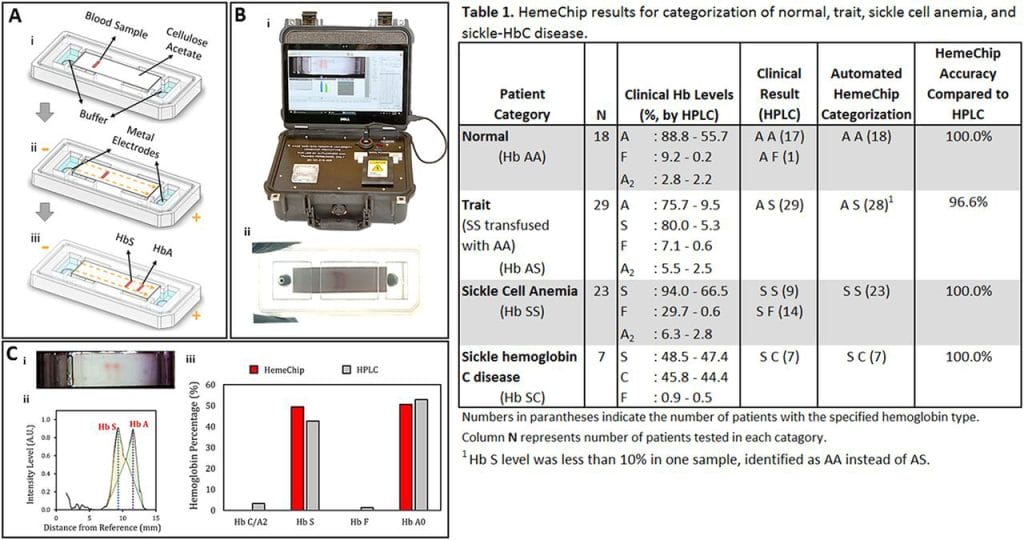
Image Credit: ScienceDirect | HemeChip Overview & Clinical Testing Results (2017)

HemeChip Technology

Navigating medical device development requires expert support at every stage to drive innovation from concept to production. Regardless of where you are in your product lifecycle, improve your speed to market with AC’s engineering teams in Research & Development, Product Development, Hardware Testing, and Manufacturing Automation.
Whether you’re a MedTech enterprise looking for targeted engineering solutions or a startup ready for full lifecycle expertise, Andrews Cooper has experience solving those “impossible” challenges. How we can we accelerate your MedTech device development?
Looking for more on MedTech engineering solutions? Explore these posts: