

Bringing a medical device from concept to market is a complex process. Beyond a great idea, it requires expert engineering, rigorous validation, regulatory compliance, and precision manufacturing—all while adapting to compressed timelines. For innovators, MedTech development hurdles can feel overwhelming without the right support. Adding multidisciplinary engineering expertise to your team is essential to eliminate roadblocks and accelerate your journey to market.

In this series, we address four critical questions for MedTech developers:
This blog—the second in our series on Accelerating MedTech Development—helps to answer the second question above. We look at how test engineering integrated into the product development lifecycle, actively preserves design integrity, ensuring product standards remain uncompromised despite complex design and regulatory demands.
MedTech devices are required to deliver consistent performance, adhere to stringent regulatory standards, and function reliably in real-world environments—all while maintaining cost-efficiency and timely market entry. Here, design integrity refers to a device’s ability to fulfill its intended purpose consistently, delivering dependable outcomes while meeting safety standards. Compromising design integrity at any point can delay regulatory approval or lead to costly recalls.
To mitigate these risks, test engineering provides a structured, proactive framework to identify and resolve issues early, strengthening a product’s reliability and compliance.

Integrating test engineering early in the design process establishes a proactive strategy to safeguard design integrity, ensuring that critical design elements are protected from the outset. For complex or high-risk MedTech devices, embedding test engineering into the Design Failure Mode and Effects Analysis (DFMEA) process enables targeted, risk-based testing aligned with potential failure modes. This approach not only strengthens device reliability but also ensures compliance with regulatory standards.
By incorporating test protocols from the beginning, teams can set baseline performance metrics, conduct stress testing, and validate that initial design choices align with both regulatory requirements and user needs. Early testing insights refine design parameters and shape downstream testing, creating a cohesive development process that reinforces product quality, reliability, and compliance.
When integrated early in the design phase, test engineering focuses on addressing the highest-priority risks identified through a comprehensive risk assessment. For instance, design engineers may need to address specific concerns with electronic sensors, signal quality, environmental durability, or long-term usability to meet stringent regulatory requirements.
By working closely with the design team, test engineers establish a framework for targeted testing across the product development lifecycle, reducing the risk of late-stage failures and enhancing device resilience, compliance, and patient safety.


Verification and Validation (V&V) are core processes that confirm a medical device meets design specifications (Verification) and fulfill real-world user needs (Validation). Test engineering supports V&V by developing and executing targeted tests that assess device safety, functionality, and compliance at each stage, ensuring that every design element aligns with regulatory and operational requirements.
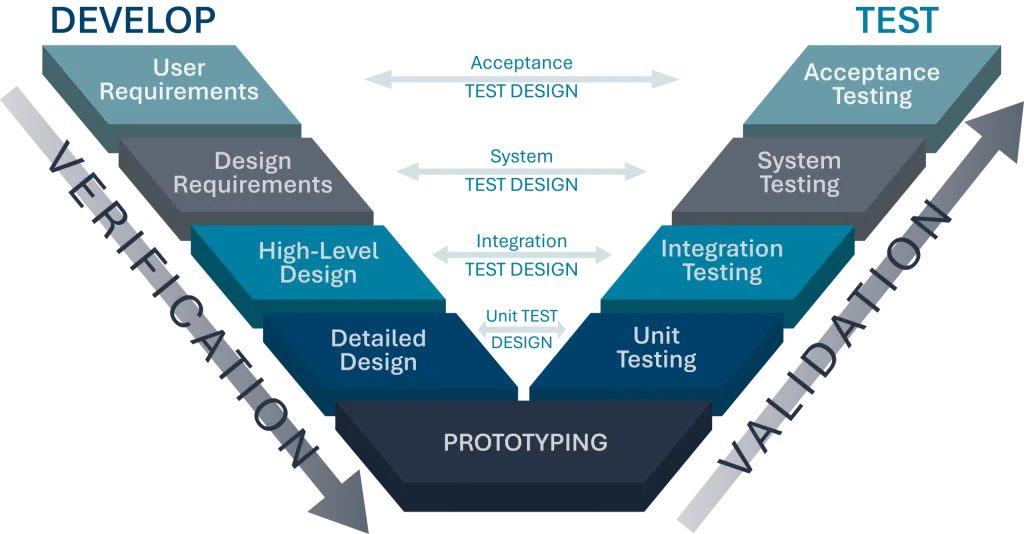
This V&V approach aligns testing with development phases, facilitating continuous refinement while reinforcing design integrity. Testing checkpoints across Unit, Integration, System, and Acceptance Testing levels enable early issue detection, incremental improvements, and risk mitigation at each development stage, guiding the product toward reliable market readiness.
Compliance with FDA, ISO, and IEC standards is essential for medical devices to ensure safety, efficacy, and reliability. Test engineering plays a crucial role in meeting regulatory requirements, such as:
Standards like these form the regulatory foundation for medical device test engineering, ensuring that each protocol and test outcome meets rigorous safety, usability, and functionality criteria.
Strategic test engineering supports Process Failure Mode and Effects Analysis (PFMEA) in manufacturing by supplying essential data to identify and reduce risks in production. This approach helps preserve the design’s integrity through to the final product, minimizing variability and non-compliance.

By working closely with the design team, test engineers establish a framework for targeted testing across the product development lifecycle, reducing the risk of late-stage failures and enhancing device resilience, compliance, and patient safety.
Test engineering is more than a verification step; it’s a proactive approach to sustaining MedTech design integrity. By integrating testing from the beginning and maintaining rigorous protocols throughout development, teams can produce compliant, reliable devices optimized for real-world use.
Discover strategic testing solutions that support your product’s journey from concept to market, ensuring safety, compliance, and lasting reliability.